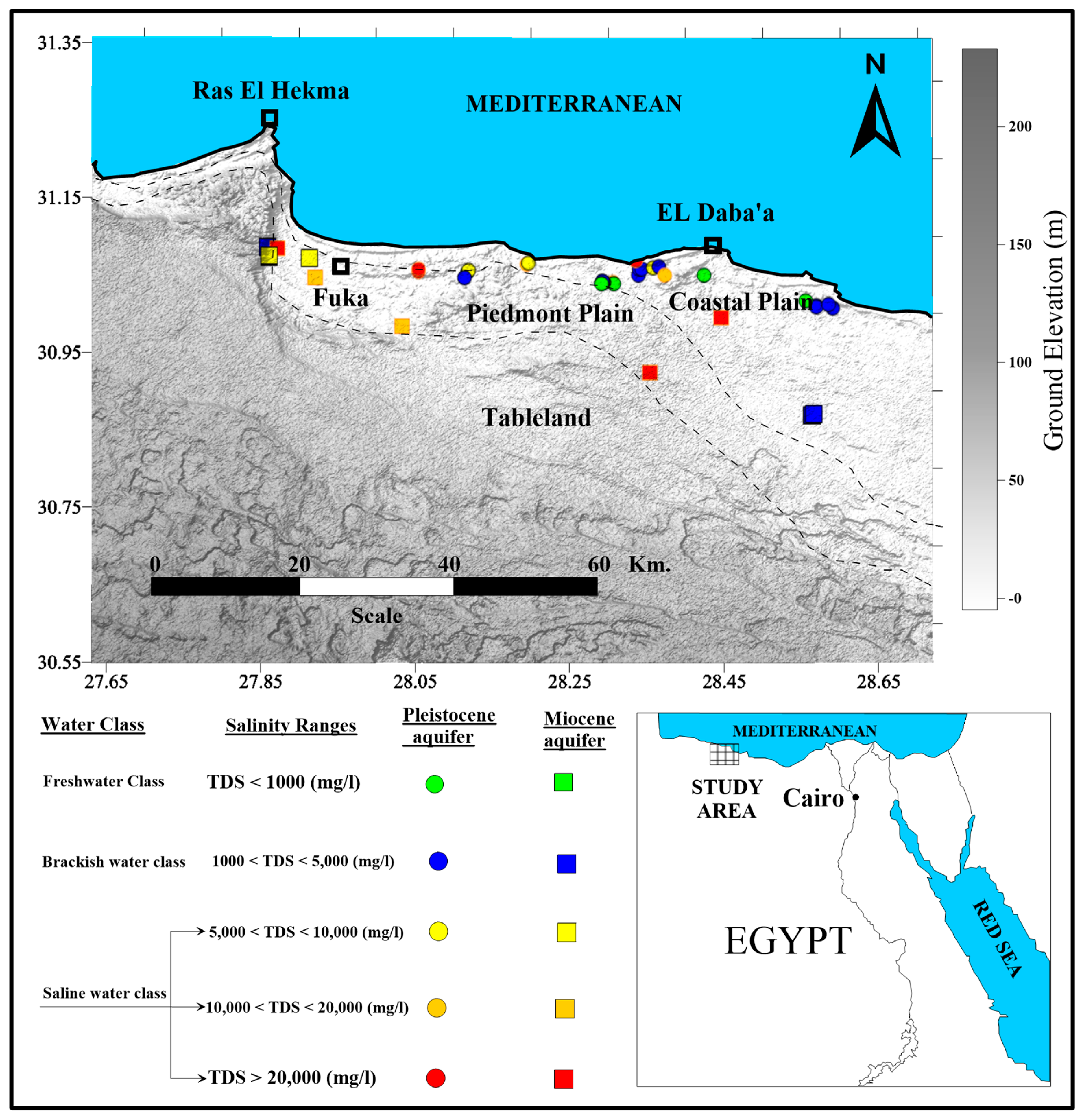
One could argue that the Lanthanides, beginning with element number 58, should be listed in the sixth row following element number 57, La. These elements are shown in a separate row called the "Lathanide Series". Before filling the rest of the 6p orbitals, we must first fill the 4f orbitals.
As the atomic number increases, the number of protons, neutrons, and electrons in each subsequent element increases, and thus the mass of the element also increases.For example, Carbon-12 has 6 protons and 6 neutrons, Carbon-13 has 6 protons and 7 neutrons, and Carbon-14 has 6 protons and 8 neutrons. An element is defined by the number of protons, but can have differing numbers of neutrons, resulting inĭifferent isotopes. The total atomic weight of the element is equal to the sum of the weights of the nucleons (protons and neutrons) and electrons.
The atomic number indicates the number of protons in the nucleus.Hydrogen (H) has atomic number 1, Helium (He) has atomic number 2, etc. Each element in the table has an assigned atomic number.The following notes were taken by me while listening to \ Step fourĬalculate the mass of reactant needed using the moles calculated in step 3 and the gram formula mass of the reactant.Click on any of the squares above for more information on an element. Moles can be compared but masses cannot so the following equation is used to help convert between moles and mass:

Calculating massīeing able to calculate masses based on balanced equations is very important. The co-efficients in balanced equations represent the number of moles reacting and being produced, it is rare that this exact ratio will be used in a reaction.įor example, some reactants can be very expensive so the experiment may be scaled down, usually one reactant is in excess or perhaps the chemist knows approximately how much product they need. Balanced equations are used to allow chemists to calculate how much product they will produce from their reactants.
